Ozone Library
All About Ozone Systems
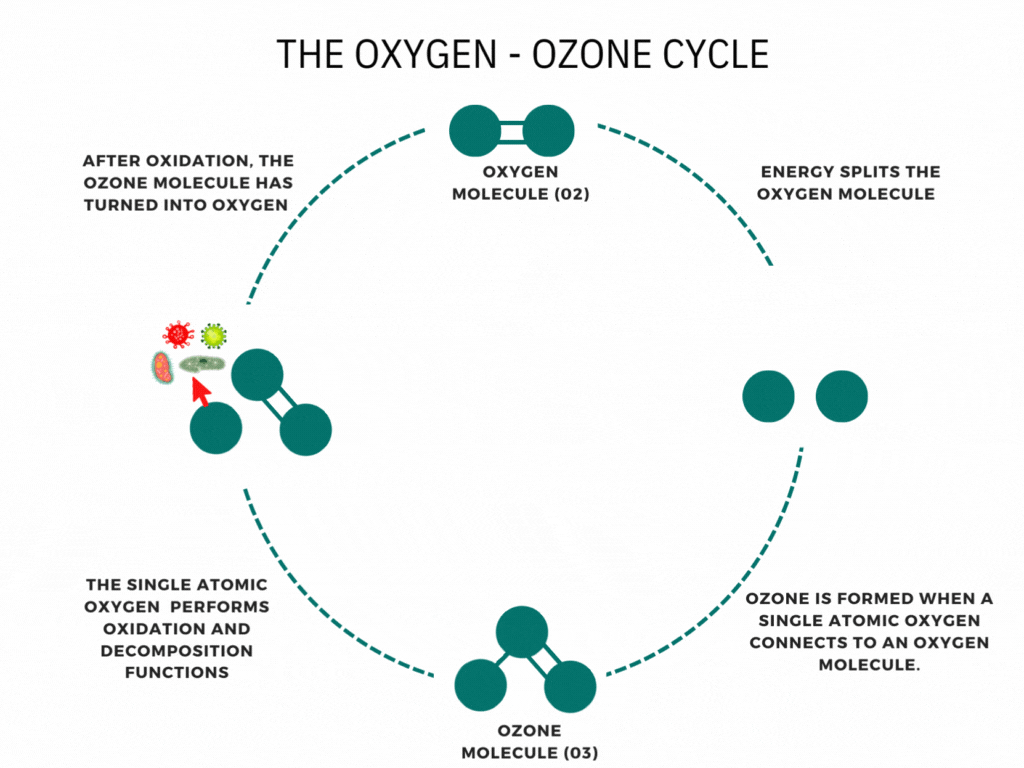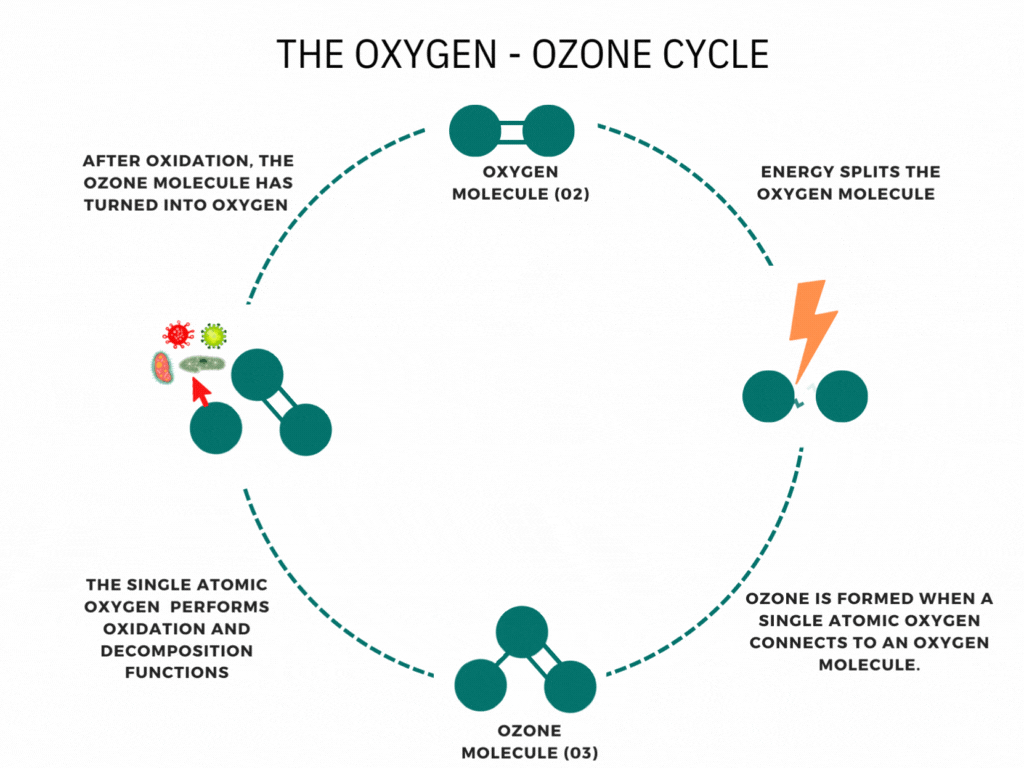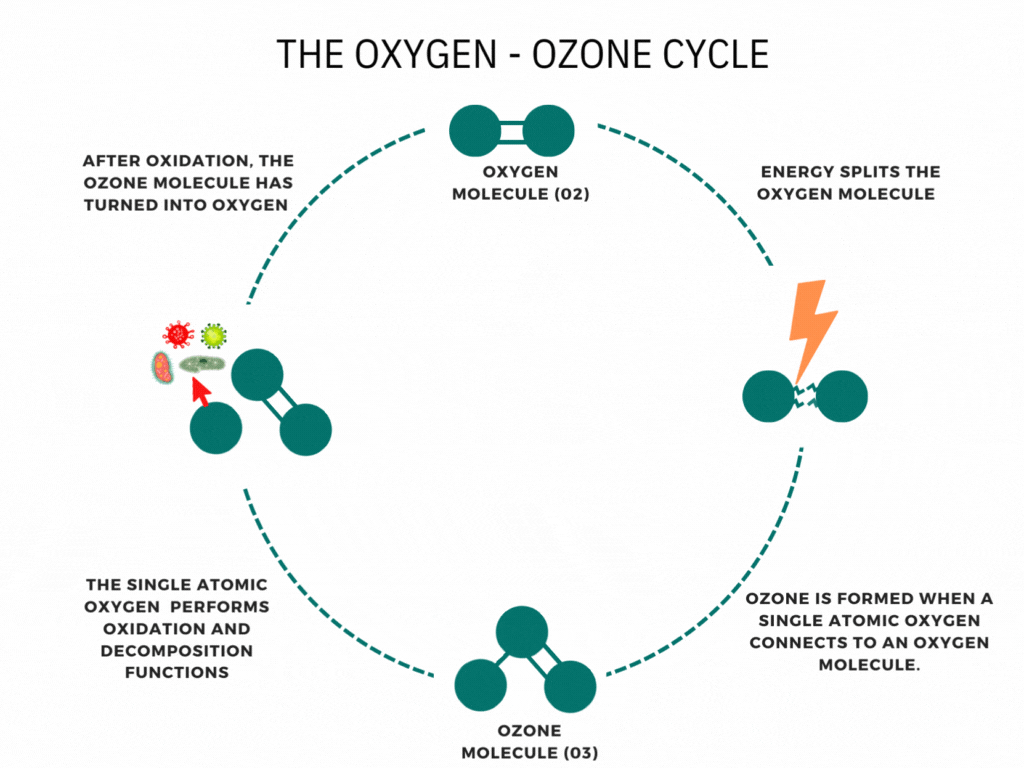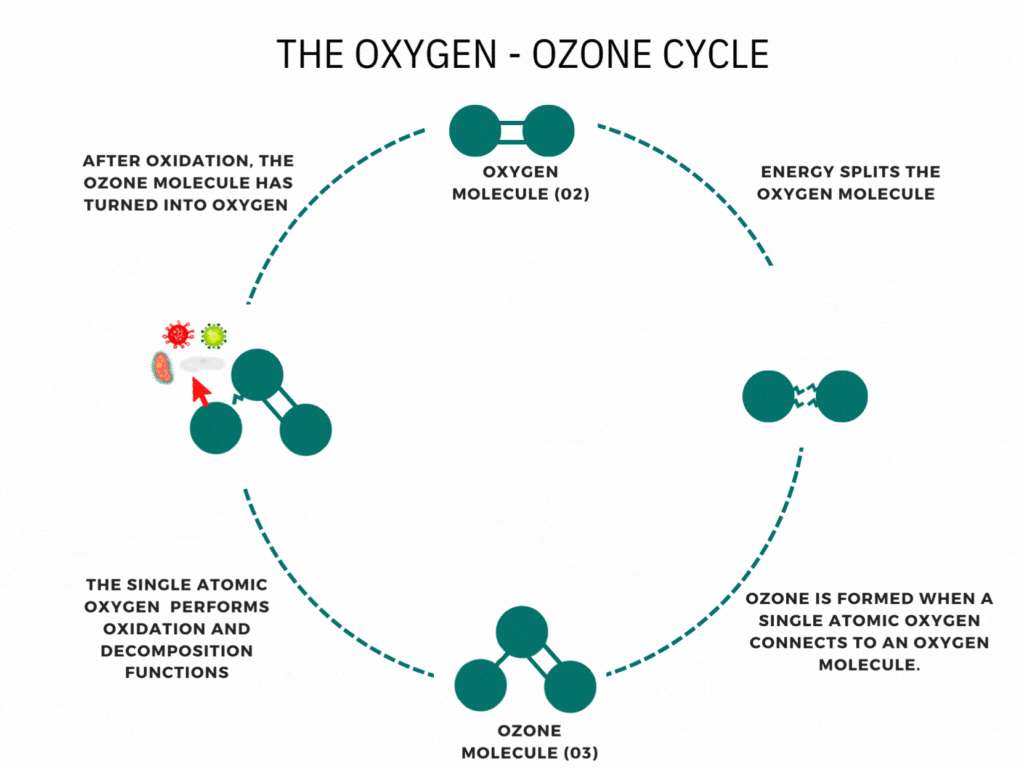
An ozone generator or machine creates ozone by supplying energy to oxygen molecules (O2). This energy causes the oxygen atoms to momentarily break apart and then reunite with other oxygen molecules to form Ozone (O3).
Ozone generators are crucial in many industrial applications because they provide an affordable and effective solution for many industrial uses due to their capacity to produce ozone gas. This potent oxidant may destroy bacteria, viruses, and other dangerous pollutants.
Whether you need an industrial ozone generator for ozone water treatment, a commercial ozone generator for industrial air purification, control indoor air pollution, or an advanced oxidation system, Ozone generators are the perfect solution.
What is Ozone?
Ozone is nature’s way of cleaning the environment. When you hear the news about the damage to the ozone layer, you may think that ozone is air pollution or smog. It is neither. Ozone is a bluish water-soluble gas with a characteristic odor. This molecule of three oxygen atoms forms when Oxygen and ultraviolet light interact high in the stratosphere; Furthermore, Waterfalls, the ocean’s surf, and thunder and lightning storms create ozone. It is the fresh smell of air at the seashore or the sweet smell after an electrical storm. The ability of ozone to emit a single oxygen atom is critical. Ozone combines with the lower atmosphere’s toxic chemicals given off by automobiles, trucks, buses, utility industries, and other poisonous gases to neutralize them. Ozone is nature’s way of cleaning our environment.
How effective are ozone generators?
It is very likely that your first online search for “ozone generators” will lead you to a series of air-feed generators. Most of these generators are sold as air purifiers or air cleaners for home use, odor removal, or indoor air cleaning. Still, they are not practical for industrial applications and water treatment.
An ozone air purifier is typically a small, portable device that produces ozone at low concentrations to clean the air in a room or small space for specific purposes, such as removing pet odors or cigarette smoke.
On the other hand, an industrial ozone generator is a larger and more powerful machine used for various industrial applications. They can produce higher ozone concentrations and are often used to treat water or remove contaminants from industrial processes.
Various industries widely use Industrial ozone generators. In the food processing industry, for example, ozone generators are used to decontaminate and sanitize food products, ensuring that they meet strict health and safety standards. The water treatment industry uses ozone generators to purify water by eliminating bacteria and viruses, improving its taste and odor, and making it safe for consumption. Pharmaceutical manufacturing uses ozone generators to sterilize equipment and surfaces, helping to prevent contamination and ensure product quality.
How Does An Ozone Generator Work?
Have you ever wondered how ozone generators work? Ozone generation uses the Corona discharge principle. It recreates lightning strikes in a controlled setting. This principle has different names depending on types and styles (dielectric barrier discharge, corona discharge, plasma block, silent corona, etc.) but is fundamentally the same. It is simply a diffuse electrical discharge through a dielectric material. The electric charge applied to the electrodes causes some oxygen molecules to split apart and temporarily combine with other oxygen molecules making a molecule of three oxygen atoms (O3).
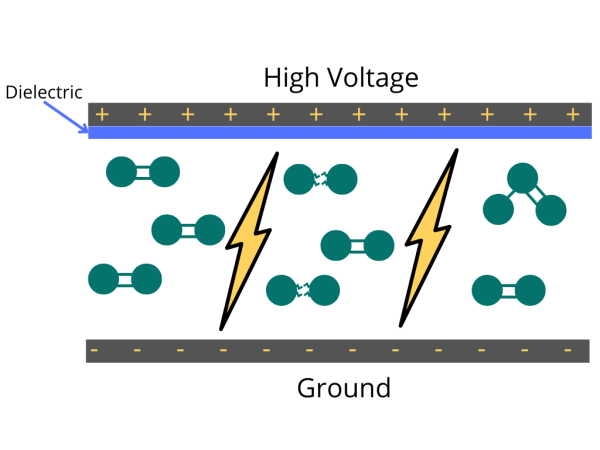
Concentrated Oxygen for Feed Gas
One of the first questions we often face from clients designing an ozone system is whether the ozone generator requires concentrated Oxygen or simply clean, dry air. I will explain why oxygen-feed ozone generators are the best choice for all industrial ozone generators.
Once we understand how ozone generation works, the problem with air-fed generators becomes clear. Only 21% of air feed is Oxygen. This low oxygen concentration significantly affects the ozone concentration that a generator can achieve. Unsurprisingly, uncommon measurement units hide their low performance (like micrograms mg/h instead of g/h). In addition, the remaining 79% of the nitrogen and other gases inside the cells will form nitric oxide deposits. In this way, ozone production will gradually decrease after a few months. Also, you will require an electrode cleaning/replacement generating an additional unnecessary expense for customers.
Even though concentrated oxygen systems are more expensive than dry air systems due to the need for a compressor and complexity, You will recover the initial investment quickly. Concentrated Oxygen refers to an oxygen supply that is at least 90% pure, with moisture removed to a dew point of -100° F. An industrial ozone generator requires this high concentration source of Oxygen to produce high levels of ozone generation.
The advantages of using concentrated Oxygen are numerous. It allows for consistent ozone output over time, and virtually all moisture is removed from the Oxygen, resulting in a more reliable ozone output and eliminating the need for corona cell maintenance. In addition, ozone concentration is higher than in dry air generators, and higher concentrations make it highly soluble in water.
Ozone For Effective Disinfection
Ozone is a powerful oxidizing agent that effectively inactivates many microorganisms, including bacteria, viruses, fungi, protozoa, and volatile organic compounds. Ozone treatment works by disrupting the cell membranes and damaging the cellular components of microorganisms, leading to their inactivation or death.
One of the critical advantages of ozone treatment is its broad-spectrum antimicrobial activity. Ozone can inactivate a wide range of microorganisms, including both enveloped and unenveloped viruses, gram-positive and gram-negative bacteria, and various types of fungi and protozoa. This characteristic makes it a versatile tool for disinfection and sterilization in multiple settings, including water treatment, food processing, and healthcare facilities.
Ozone treatment is particularly effective against waterborne pathogens, including Cryptosporidium and Giardia, which can cause gastrointestinal illness. Ozone treatment is highly effective in inactivating these pathogens, even at low doses. In addition, ozone treatment controls biofilm formation and mold spores, which can be a constant source of contamination in water distribution systems.
In healthcare settings, ozone treatment controls indoor air contaminants and the spread of infectious diseases, including mold spores, bacteria and viruses such as SARS-CoV-2. Ozone disinfect surfaces, equipment, and air pollutants in hospital rooms and other healthcare settings, reducing the risk of healthcare-associated infections.
Are ozone generators safe?
Using ozone generators correctly ensures their safety. Ozone can react with and eliminate many contaminants, including bacteria, viruses, and odors. However, Ozone can also harm humans and animals if exposed to it at high concentrations for extended periods.
Exposure to high Ozone levels can cause respiratory irritation, coughing, chest pain, and shortness of breath. Also, prolonged exposure to high ozone levels can damage the lungs and cause severe health problems. Additionally, Ozone can also be explosive in certain conditions. If you take proper safety precautions, you can safely use Ozone.
Following safety guidelines and regulations is essential when working with Ozone. These guidelines include using personal protective equipment, ensuring proper ventilation, and monitoring ozone levels. It is vital to follow the manufacturer’s instructions for safe device usage and to keep ozone concentrations in the air below recommended limits.
Ozone Applications
Industrial Ozone generators offer a modular design that is suitable for a range of small, medium, and large ozone applications. Ozone generators have become a key tool for ensuring the safety and quality of industrial processes and products, including but not limited to the following:
- Ozone drinking water treatment
- Ozone bottled water
- Ozone wastewater treatment
- Industrial Disinfection
- Ozone air purification and odor control
- Ozone soil remediation
- Barrel sanitization in wineries
- Pharmaceutical processes
- Ozone for Laboratories and oxidation process
- Ozone laundry
- Semiconductor and atomic layer deposition
- Ozone mold remediation, and more.
“Aqueous vs Gaseous Ozone: Understanding the Differences”
Ozone generator systems can be classified based on their application form, whether aqueous or gaseous ozone. Aqueous ozone generators produce ozone in a liquid or water-based environment, typically using an ozone diffuser to dissolve ozone into the water. People commonly use aqueous ozone for water treatment, disinfection, and cleaning. It is crucial to follow appropriate usage guidelines to ensure safe and effective results.
On the other hand, gaseous ozone is directly released into the ambient air, or used in a system for sterilization, disinfection, and odor control applications. The choice between aqueous and gaseous ozone generators depends on the intended application.
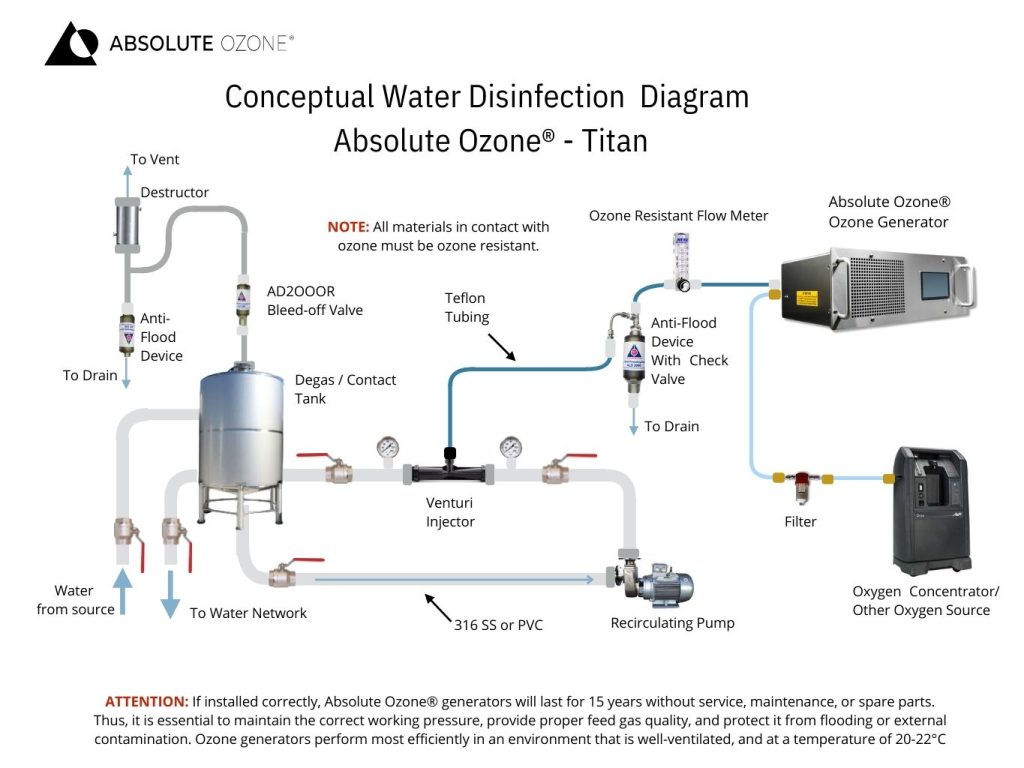
Aqueous Ozone Systems (Ozonization)
Ozone gas is partially soluble in the liquid. However, it is possible to make ozone machine reach high mass transfer efficiency using proper methods and equipment. Currently, venturi-based mass transfer systems are the preferred method for dissolving ozone in water due to their simplicity, economy, and effectiveness. The resulting ozone-infused water is then run through a reaction chamber where it comes into contact with contaminants, effectively destroying them.
This process leaves behind clean, purified water safe for consumption and use. In addition to its disinfectant properties, ozone also helps to reduce odors, tastes, and colors in the water, making it a versatile and efficient water treatment solution.
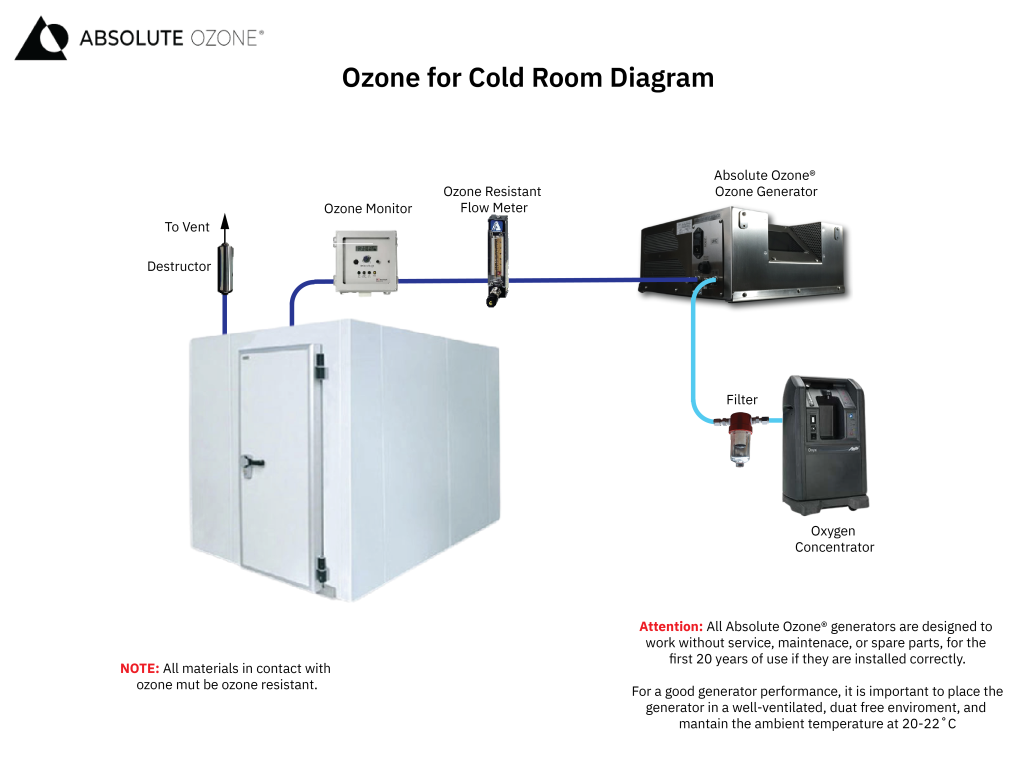
Gaseous Ozone Systems
The industry uses gaseous ozone in a wide range of air treatment and odor mitigation applications, such as controlling indoor air pollution, cold room disinfection, and biofuel odor control. Ozone is circulated through the air to treat any airborne particles, contaminants and pollutants that may be resting on exposed surfaces. You can efficiently reduce or completely remove airborne odors and contaminants using ozone and its oxidation process.
It is essential to follow safety guidelines and regulations of gas ozone systems.
TESTIMONIALS
HERE IS WHAT SOME EXPERTS SAY ABOUT OZONE:
“The trouble is that one soon gets used to dirty air, and offensive odors are not apt to be noticed after a while, but the fact remains that pure air is more wholesome than contaminated air. In daily life, it is almost impossible to provide fresh air. No amount of ventilation, not even an unbearable draft, will be able to keep a room or a place in proper condition unless one takes recourse to ventilate with ozonized air. Removal of foul odors by means of air flushing is an absolute impossibility, and yet they should be removed. That is where ozone comes in. The method of purifying the air by ozone has the advantage of being fully reliable, very efficient, and inexpensive.”
• A. Vosmaer, Ph.D., Electrical and Chemical Engineer, London, England •
In “Ozone, its Manufacture, Properties and Uses”
“Ozone destroys virtually all odors that are present. It does not merely mask them. The destruction of odors is impossible when air is circulated only, or when oxygen is used. Cold storage warehouses, which store all kinds of food products, have proven this. Odors are not present regardless of how strong they might be, or where they may originate if only low concentrations of Ozone are used.”
• E. W. Reisbeck. M. E., Ozone Research Authority •
In “Air Conditioning and Ozone Facts”
“As a deodorant for odors and stenches of organic origin, ozone has long proven effective and we can only confirm this general opinion.”
• DR. PHILIP DRINKER, SCHOOL OF PUBLIC HEALTH, HARVARD UNIVERSITY
“Ozone treatment destroys organic odors. Ozone is a deodorizer of powerful stenches, such as from garbage incineration and fat rendering. When the odors from chimneys cause public nuisance, Ozone has big commercial potential.”
• Milton J. Rosenaw, MD
In “Preventive Medicine and Hygiene”
“Experiments with cholera and typhus bacteria are rather awkward to be carried out in a private plant, handling, say a million gallons of water per day, and the firm Siemens and Halske were very fortunate to find the Prussian State officials willing and ready to test the matter. Dr. Ohlmueller and Dr. Prall published the results of their findings regarding the action of Ozone on bacteria. The experimental series covered the effect of Ozone on pure water infected with 16,000 cholera, at another time with 30,000 to 40,000 typhus, and another time with 20,000 to 40,000 coli bacteria. The result was absolute sterility after treatment. We then tested infected ordinary river water that contained over 4,000 bacteria. After treatment with Ozone, some 5 or 6 were left, and those were harmless.”
• E. HOWLETT, ME
“Ozone treatment is a powerful germicidal. Its high germicidal activity is doubtless due to its oxidizing power.”
• E. K. RIDEAL, PH.D.
“Ozone generators have been installed in many homes, and that super-oxygen is particularly destructive to all microbes and at the same time it makes inert the dangerous dust with its bacteria-laden tenants.”
• W. E. ANGHINBAUGH, MD •
“One part Ozone in 2 million p. solution renders the virus poliomyelitis inactive within 2 minutes compared with the double amount of chlorine using 3 hours.”